Embryonic Eye Development
By Linda Conlin, BS, ABOC, NCLEC
Release Date: November, 2012
Expiration Date: September 28, 2017
Learning Objectives:
Upon completion of this program, the participant should be able to:
- Gain knowledge of the anatomy and physiology of the eye through an understanding of its embryonic development and tissue specialization.
- Understand how infants see based on the development of the visual system and common refractive errors present at birth.
- Gain knowledge of the vision conditions in infants and options for vision correction.
Faculty/Editorial Board:
 With over 30 years of experience and licensed in Connecticut, Massachusetts and Rhode Island, Linda Conlin is a writer and lecturer for regional and national meetings. She is chair of the Connecticut Board of Examiners for Opticians and is a manager for OptiCare Eye Health and Vision Centers, a multidisciplinary ophthalmic practice in Connecticut.
With over 30 years of experience and licensed in Connecticut, Massachusetts and Rhode Island, Linda Conlin is a writer and lecturer for regional and national meetings. She is chair of the Connecticut Board of Examiners for Opticians and is a manager for OptiCare Eye Health and Vision Centers, a multidisciplinary ophthalmic practice in Connecticut.
Credit Statement:
This course is approved for one (1) hour of CE credit by the National Contact Lens Examiners (NCLE) Course CJMI036-1 and the American Board of Opticianry (ABO) Course CWJM030-1 & SWJM266-2.
When working with infants and very young children, eyecare professionals have the opportunity to set the course for how these tiniest of patients see for the rest of their lives. Knowing how the eye develops can ensure that children enjoy the beauty of a sunset or the words of a novel. The ability to see is an amazing journey from fertilization to the first months of life. Any interruption or misstep along the way will result in eye and vision problems.
A BIT OF HISTORY
Scientists have studied the complex process of human eye development for nearly two centuries. It started in 1817, when Christian Heinrich Pander determined that the optic vesicle derives from the fore-brain. Thirteen years later, Emil Huschke established that the crystalline lens develops from surface ectoderm cells, and in 1835 that the optic cup is formed by the optic vesicle. Between 1850 and 1855, Robert Remak verified that the lens epithelium and fibers develop froFm the lens vesicle. In the latter half of the 19th century, information about retinal development emerged. In 1861, Rudolph Albert vonKolliker determined that the retina forms from two layers of the optic cup, and in 1875, Johannes Müller established that the optic nerve fibers begin in the retina and grow into the brain to make vision possible.
EARLY DEVELOPMENT
Since Müller's time, science has uncovered a great deal more about the eye's transition during gestation. Let's start shortly after fertilization as the embryonic cells divide quickly going from two cells to 128 cells, which is called a blastula. The blastula has a spherical layer of cells called the blastoderm. The blastoderm consists of three layers—the endoderm, meso-derm and ectoderm—from the inside to the outside. The cells of each layer have distinctive characteristics that will enable the cells to develop into specific types of tissue. While we will examine only the eye, keep in mind that the same three embryonic layers give rise to all of the varied tissues in the body.
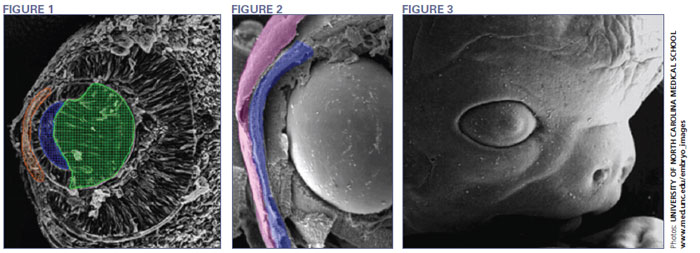
The human eye begins to develop during the 17th day of gestation. Mesoderm cells, the middle layer of the blastoderm, and ectoderm cells, the outer layer of the blastoderm, form the eye fields in the neural area of the embryo. Optic vesicles develop in the eye fields and in five days, infold to form the optic cup. At this point, the retina and crystalline lens begin to develop. Meanwhile, surface ectoderm cells are becoming thicker. The lens forms from these thickened cells. By the 32nd day, you can easily identify the lens, and during the next three to three and a half weeks the lens will grow to the size it will be at birth (Fig. 1).
Between 30 and 35 days, you can see the start of the iris. In two more weeks, it is fully developed. While the iris grows, the optic stalk, the precursor to the optic nerve, forms a critical connection to the forebrain at 36 days.
Eye development during the first trimester of pregnancy is like watching the grand finale of Fourth of July fireworks. Cells and tissue develop quickly and simultaneously to form various eye structures. For example, as the lens is develop ing, the cornea forms from ectoderm cells covering the lens. At about five weeks of pregnancy this tissue is two cells thick. What occurs over the next two weeks is amazing. The number of ectoderm cells will nearly double, creating the corneal epithelium (Fig. 2). At this point, the lens has completed its formation and separates from the ectoderm. This separation creates the anterior chamber. Meanwhile, neural crest cells have separated from the neural plate and migrate to the anterior chamber to form other neuronal and non-neuronal cells. Once separated, they move into the area between the lens and the corneal epithelium, forming the corneal endothelium and stromal keratocytes, the primary cells of the stroma. To all that activity, add the formation of the orbits and extraocular muscles at four weeks gestation. Depending on eye growth after birth, however, the orbits may not mature until adolescence.
During the sixth week the lacrimal glands begin forming but produce no tears until the third month after birth, which is why infants shed no tears when they cry. At eight weeks, the eyelids start to form and fuse together protecting the other developing eye structures (Fig. 3). During the fifth month, a two-month process begins—the separation of the eyelids. While the eyelids are fused, the corneal epithelium decreases back to two cells in thickness and enlarges and matures. The maturing, highly-hydrated cornea does not become transparent until the eye becomes functional in the seventh month. Descemet's membrane matures just prior to the eyelids opening. Bowman's membrane develops in the fifth month as the cornea becomes inervated.
Although the iris begins to develop from the rim of the optic cup at five weeks, the muscles controlling the aperture do not develop until five months. Since constriction of the pupil in response to light is not necessary in the darkness of the womb, it doesn't begin until the eighth month. It takes another two weeks for this response to become consistent. Because this reflex develops late in gestation, babies born prior to 34 weeks gestation need eye protection. In a neonatal intensive care unit, for example, premature babies wear eye covering. In the 1970s and 1980s, the medical community believed that stimulating visual attention, focus and tracking was critical to the visual development of premature infants. Recent research, however, shows that because the light control response is underdeveloped, the need for protection from light outweighs the need for visual stimulation until the premature infant has reached the point in development that would have been complete at 40 weeks gestation.
At seven weeks, the sclera develops from embryonic tissue, which allows the formation of blood vessels. The cornea connects to the sclera which is nourished by blood. But why then does the cornea lack blood vessels? The answer is that the cornea develops from ectoderm rather than embryonic tissue. The ectoderm cells provide a clear medium for light, and the lack of blood minimizes tissue rejection in corneal transplants. It is for this very reason that the cornea forms from the same embryonic cells as the crystalline lens. How's that for efficiency? In addition, we also know that both tissues have refractive power.
During this time the retina is still evolving. The neural cells of the retina develop from a single embryonic layer. The cells then dif ferentiate into specific types according to their projected role in the visual process, such as rods and cones, with axons from the retinal ganglion cells forming the optic nerve. It takes six months for the retinal layers to grow from the neural ectoderm. The macula needs four or five months just to begin, and it will mature six months after birth. As the retina and lens develop, the vitreous forms between them. In the meantime, the neural connections between the eye and brain have been developing, taking five months to complete.

The bulk of the highly complex development of the retina occurs between 24 weeks gestation and 3 to 4 months of age, when the optic nerve becomes fully myelinated. Rod cells are better developed at birth resulting in scotopic vision in infants. As a result, newborns see primarily dark, light and shades of gray. Color discrimination occurs at 3 months of age when the cones mature.
Between 28 and 30 weeks gestation, the fetus develops rapid eye movement and sleep patterns. What does that have to do with vision? The connection between the retina and the brain is the essence of vision, and REM and sleep cycles help messages from the retina synchronize with the brain waves of the visual cortex. If these cycles are disrupted or the infant is deprived of sleep, it can significantly interfere with visual development.
FROM BIRTH TO BEYOND
Even after nine months of simultaneous, rapid-fire tissue formation, the eye is not mature at birth. A newborn's eye is about two-thirds (65 percent) of its full size, so the eye must undergo a series of changes that can take as long as a year. The changes include growth of the orbit, changes to the crystalline lens and final maturation of the macula, as well as pigmentation of the iris. Horizontal, vertical and rotational eye movements are somewhat uncoordinated and develop at different rates. On the neural level, receptive fields of the retinal ganglion cells are smaller in infants. Many cells in the lateral geniculate nucleus (LGN), part of the pathway between the retina and the visual cortex, do not yet respond to visual input. Because of an undeveloped ability to accommodate, visual acuity in infants is about 20/400 which results in a focal distance of 8 to 10 inches. Acuity improves to 20/25 at about 6 months of age. This process is called emmetropization.
Corneal curvature also changes after birth. Average keratometry readings in infants are about 54D on the first day of life and flatten to about 48D at 1 year of age. Moreover, the brain's accurate interpretation of visually transmitted images seems to take a little practice. Spatial orientation of images takes even longer because stereoscopic vision doesn't fully develop until 15 to 18 months of age.

It is normal to find low levels of hypero-pia in infants. Hyperopia greater than 3D occurs in 25 percent of newborns. Refraction of 4D is found in 9 percent of infants at 6 months. By 7 to 9 months it falls to 5 percent and continues to decline to 3.6 percent at 1 year of age. As can be expected with such rapid changes in corneal curvature, astigmatism, usually with the rule where the vertical meridian is steeper, is higher in the first year of life. The incidence of astigmatism decreases from 1.5 to 3 years of age as the cornea flattens.
Instances of anisometropia, different refractive errors in each eye, are common at birth but decrease quickly with age. If all the components of the eye grow proportionally, any refractive error decreases. Myopia, however, seems to rebound. As is the case with hyperopia, infants born myopic will become less so. However, the tendency is for myopia to increase into the higher ranges once the child begins school.
WHEN NATURE ISN'T ENOUGH
While the majority of refractive errors present at birth will naturally correct themselves as the child grows, some conditions require the intervention of an eyecare professional. Congenital cataracts occur in 1.7 of 10,000 births and are the most common cause of treatable childhood blindness. Congenital cataracts can be bilateral or unilateral and are caused by genetics, metabolic disorders, trauma and maternal infection. Surgical treatment must occur before the child is a year old to preserve visual development. For post-surgical vision correction, the use of intra-ocular lenses in infants is not unheard of, but it is controversial. Spectacles may be used, but they are impractical for a small child, and the high power lenses can cause distortion and problems with image size, especially aneisikonia in the case of unilateral cataract. The most common vision correction is a contact lens or lenses fit seven to 10 days after surgery.

Retinopathy of prematurity occurs in 16 percent of premature births, usually prior to 30 weeks gestation and in infants weighing less than 3 pounds. As we know, retinal development is incomplete at this point, and visual stimulation that occurs too early may initiate abnormal blood vessel growth in the retina. This condition is usually accompanied by high myopia. Amazingly, most infants recover from this condition naturally, though some may continue to need correction for myopia. In about 10 percent of cases however, laser therapy to stop blood vessel growth or surgery for a detached retina is necessary. Another cause of myopia in infants is an interruption in the visual pathway that results in increased eyeball growth in an attempt to compensate for the poor visual images the brain receives.
Although low to moderate levels of hyperopia are normal at birth, infants with hyperopia in excess of 3.50 diopters are more likely to remain hyperopic. Hypero-pia at this level is usually the result of developmental problems in utero. It may be accompanied by against the rule astigmatism in which the horizontal meridian is steeper. Infants born with high levels of hyperopia are more likely to develop strabismus and amblyopia as well. Eyecare practitioners are hesitant to prescribe early vision cor rection because it may interfere with emmetropization. However, spectacle correction used part of the time may reduce the incidence of strabismus and amblyopia.
 As mentioned previously, it is normal to see an infant's eyes misaligned at first because of immature focusing and muscle control. If the eyes remain misaligned after 4 months of age, it may be an indication of strabismus. Because the brain is not receiv ing good messages from the turne d eye, amblyopia can result. As many as 5 percent of preschool-aged children have amblyo-pia, so it is clear that early intervention is important to preserve visual development. Spectacles with an occluder or frosted lens over the normal eye are used to stimulate visual development in the affected eye. When spectacles are impractical for an infant or an active toddler or preschooler, an occluder or high power "fogging" contact lens can be used. Contact lenses offer the benefit of staying in place but require that caregivers are trained to apply, remove and clean the lenses.
As mentioned previously, it is normal to see an infant's eyes misaligned at first because of immature focusing and muscle control. If the eyes remain misaligned after 4 months of age, it may be an indication of strabismus. Because the brain is not receiv ing good messages from the turne d eye, amblyopia can result. As many as 5 percent of preschool-aged children have amblyo-pia, so it is clear that early intervention is important to preserve visual development. Spectacles with an occluder or frosted lens over the normal eye are used to stimulate visual development in the affected eye. When spectacles are impractical for an infant or an active toddler or preschooler, an occluder or high power "fogging" contact lens can be used. Contact lenses offer the benefit of staying in place but require that caregivers are trained to apply, remove and clean the lenses.
CONCLUSION
These are only some of the more common vision problems in infants, but of course, you may see others in your practice. Considering the intricate processes of human eye development and the many chances for error along the way, vision is the most amazing of our senses. As eyecare professionals, we have the rewarding work of picking up where nature may have left off.