|
THE LATEST RESEARCH
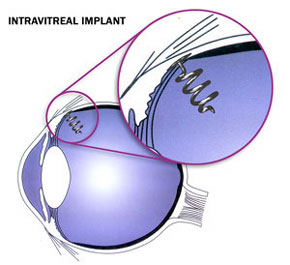
Intravitreal Bevacizumab for CNV Secondary to AMD
In a retrospective study involving 266 eyes (266 consecutive patients)
with choroidal neovascularization (CNV) secondary to age-related macular
degeneration (AMD), intravitreal injection (1.25 mg) of bevacizumab
(Avastin) decreased macular thickness and improved visual acuity with
no apparent short-term safety problems. Follow-up information was available
for 251 (94.4 percent) of the 266 eyes.
The mean central macular thickness decreased to 247 µm at one
month and 213 µm at three months from the baseline mean of 340
µm (p<0.001). Mean visual acuity at baseline was 20/184. At
one month, (n=244) mean visual acuity was 20/137 (p<0.001). Mean
visual acuity at two months (n=222) was 20/122 (p<0.001). Mean visual
acuity at three months (n=141) was 20/109 (p<0.001). The proportion
of patients whose visual acuity improved (halving of the visual angle)
was 30.3 percent, 31.1 percent and 38.3 percent at one, two and three
months respectively.
At one month, two patients had mild vitritis, as did one patient at
two months. No endophthalmitis, increased intraocular pressure, retinal
tears, or retinal detachments occurred. The risk for thromboembolic
disorders was not different than previously reported from studies of
bevacizumab and AMD.
Source: Spaide RF, Laud K, Fine HF, et al. Intravitreal bevacizumab
treatment of choroidal neovascularization secondary to age-related macular
degeneration. Retina 2006;26(4):383-390.
Studies Evaluate Repeat and Dose-Escalating Injections of Ranibizumab
Two recently published studies, one with dose escalation, evaluated
the safety and effects of repeat intravitreal injections of ranibizumab
(Lucentis) in patients with subfoveal CNV secondary to AMD.
In one of the studies (Rosenfeld et al.), which was uncontrolled, randomized,
and conducted at two centers, patients with primary or recurrent CNV
were treated with five, seven, or nine injections at two- or four-week
intervals for 16 weeks and followed through day 140 (four weeks after
last injection). Three different escalating doses, ranging from 0.3
mg to 2.0 mg, were administered. The study enrolled 32 patients; 29
received an injection at baseline; and 27 completed the study through
day 140 (week 20).
Results were similar across all three treatment groups. Inflammation
did not increase with repeat or dose-escalating injections. No significant
lesion growth occurred through day 140, and area of leakage decreased.
Median and mean visual acuities improved in all three dose groups; three
patients lost 15 or more letters. All patients experienced mild ocular
adverse events; iridocyclitis (83 percent) and injection-site reactions
(72 percent) were the most common. Transient and mild elevations of
intraocular pressure were common. No serum anti-ranibizumab antibodies
were detected.
In another study (Heier et al.), which was multicenter and controlled,
repeat intravitreal injections of ranibizumab were found to be safe
and associated with improved visual acuity and decreased leakage from
subfoveal predominantly or minimally classic CNV. Sixty-two of the 64
randomized patients completed the two-part, six-month study.
In Part One, patients were randomized to monthly injections for three
months (four injections of 0.3 mg or one injection of 0.3 mg followed
by three injections of 0.5 mg) or usual care. In Part Two, patients
could continue their regimen for three additional months or cross over
to the alternative treatment. Eighty percent of patients randomized
to 0.3-mg of ranibizumab and 79 percent randomized to 0.5-mg in Part
One continued the same treatment in Part Two. Eighty-two percent of
patients randomized to usual care in Part One crossed over to ranibizumab
treatment in Part Two.
After four injections (day 98), mean visual acuity increased 9.4 +/-13.3
letters in the 0.3-mg group and 9.1 +/-17.2 letters in the 0.5-mg group,
while it decreased 5.1 +/-9.6 letters in the usual care group. In Part
Two, (day 210), visual acuity increased from baseline 12.8 +/-14.7 and
15.0 +/-14.2 letters in patients who continued with 0.3 and 0.5 mg,
respectively. Among patients who were initially randomized to or continued
ranibizumab treatment, visual acuity improved 15 letters or more from
baseline in 26 percent of patients at day 98 and 45 percent of patients
at day 210. Areas of leakage and subretinal fluid decreased.
The most common ranibizumab-related adverse events were reversible inflammation
and minor injection-site hemorrhages. Serious adverse events included
iridocyclitis (one patient), endophthalmitis (one patient), and central
retinal vein occlusion (one patient). The proportion of patients experiencing
increased intraocular pressure, which was transient, was 22.6 percent.
Sources: Rosenfeld PJ, Heier JS, Hantsbarger G, Shams N. Tolerability
and efficacy of multiple escalating doses of ranibizumab (Lucentis)
for neovascular age-related macular degeneration. Ophthalmology 2006;113(4):632.
Heier JS, Antoszyk AN, Pavan PR, et al. Ranibizumab for treatment of
neovascular age-related macular degeneration: a phase I/II multicenter,
controlled, multidose study. Ophthalmology 2006;113(4):642.
Sequenced, Combined Treatment for RAP Shows Promise
Treatment with intravitreal triamcinolone followed in seven to 14 days
by indocyanine green angiography-guided photodynamic therapy with verteporfin
(Visudyne) reduced or eliminated cystoid edema, achieved rapid regression
of neovascularization, and stabilized or improved visual acuity in patients
newly diagnosed with retinal angiomatous proliferation (RAP).
The 27 eyes of 26 patients treated with this sequenced combination therapy,
termed pharmacology-pause-photodynamic therapy (PPP), were followed
for 12 months. Angiographic leakage completely resolved in 89 percent
of the eyes. Leakage recurred in eight eyes in three to 11 months and
completely resolved after subsequent treatment.
Visual acuity improved in 37 percent of the eyes and remained stable
in 52 percent. The mean Snellen equivalent visual acuity at baseline
as well as after triamcinolone injection before PDT was 20/150 (range
20/40 to 20/400). At six months, mean visual acuity was 20/100 (range
20/40 to 20/400). After six months, visual acuity improved by three
or more lines in five eyes (19 percent), remained stable (+/-2 lines)
in 19 eyes (70 percent), and decreased by three or more lines in three
eyes (11 percent). After 12 months, mean visual acuity was 20/100 (range
20/50 to 20/400). Visual acuity improved by three or more lines in 10
eyes (37 percent), remained stable (+/-2 lines) in 14 eyes (52 percent),
and decreased by three or more lines in three eyes (11 percent).
Source: Freund KB, Klais CM, Eandi CM, et al. Sequenced combined
intravitreal triamcinolone and indocyanine green angiography-guided
photodynamic therapy for retinal angiomatous proliferation. Arch Ophthalmol
2006;124:487-492.
Direct Injection of tPA and Air-Fluid Exchange for Macular Hemorrhage
Direct injection of tissue plasminogen activator (tPA) and air-fluid
exchange (no intraoperative clot lysis period) produced positive results
in a case series involving 17 patients with subretinal macular hemorrhages
due to AMD.
The patients underwent three-port pars plana vitrectomy to enable direct
cannulation of the subretinal space and injection of 48 µg of
tPA into the hematoma and partial fluid-air exchange. They spent one
hour after the procedure in a face-up supine position and spent the
night in an upright position.
Visual acuity stabilized or improved in 88 percent of the patients.
The hemorrhage completely cleared in nine patients and partially cleared
in eight patients. The hemorrhage recurred in two patients, and one
patient underwent a retinal detachment repair. Outcomes were similar
for predominantly classic and occult lesions.
Source: Singh RP, Patel C, Sears JE. Management of subretinal
macular haemorrhage by direct administration of tissue plasminogen activator.
Br J Ophthalmol 2006;90:429-431.
Report Number 1 from PTAMD Trial Published
According to Report Number 1 from the Prophylactic Treatment of Age-Related
Macular Degeneration (PTAMD) trial, using a subthreshold 810-nm diode
laser to treat an eye with multiple large drusen when neovascularization
from AMD has already affected the other eye increases the risk of developing
CNV in the treated eye.
The 244 patients in the multicenter, prospective, randomized, controlled
trial had a neovascular or advanced AMD lesion in one eye, and the fellow
(study) eye had at least five drusen greater than or equal to 63 µm,
best-corrected visual acuity of 20/63 or better, and no evidence of
neovascularization at baseline. Study eyes were treated with a grid
of 48, 125-µm extrafoveal laser spots delivered in a ring outside
of the foveola. Patients returned for follow-up at three, six, 12, 18,
24, 30, and 36 months. Retreatments were not permitted.
Throughout the follow-up period, the rate of CNV in study eyes consistently
exceeded the rate in observed eyes (15.8 percent vs. 1.4 percent at
one year, p=0.05). Study eyes showed a higher rate of visual acuity
loss at three and six months, but after six months, no significant differences
were observed between groups.
Source: Friberg TR, Musch DC, Lim JI, et al. Prophylactic
treatment of age-related macular degeneration Report Number 1: 810-nanometer
laser to eyes with drusen. Ophthalmology 2006;113:612.
PDT Plus Triamcinolone for Occult CNV with Two-Year Follow-Up
In a single-center, nonrandomized, prospective, interventional case
series, improvement in visual acuity was maintained for the two-year
follow-up period in most patients treated with a combination of verteporfin
PDT and intravitreal triamcinolone for occult CNV due to AMD.
Forty-one eyes of 41 patients underwent PDT and intravitreal injection
of triamcinolone (solution containing 25 mg) 16 hours later. If CNV
persisted, the procedure was repeated after three months. Thirty-four
eyes (82.9 percent) received re-treatment at three months, but no additional
re-treatments were necessary. The mean number of re-treatments was 1.8.
Mean visual acuity at baseline and three, six, 12, 18, and 24 months
were as follows: 20/133, 20/115, 20/101, 20/84, 20/83, and 20/81.
Eleven of the 41 eyes (26.8 percent) underwent cataract surgery between
six and 15 months after the first treatment. Nine patients were treated
for a transient steroid-induced increase in intraocular pressure.
Source: Augustin AJ, Schmidt-Erfurth U. Verteporfin and intravitreal
triamcinolone acetonide combination therapy for occult choroidal neovascularization
in age-related macular degeneration. Am J Ophthalmol 2006;141(4):638.
ACE Inhibitor Shown to Have No Effect on CME
The results of a 40-patient, single-center study indicated that treatment
with the angiotensin-converting enzyme (ACE) inhibitor lisinopril had
no effect on inflammatory cystoid macular edema (CME) or visual acuity.
In the randomized, double-blind study, the patients, diagnosed with
CME, received 10 mg of lisinopril or placebo daily for three months.
After a two-month wash-out period, patients crossed over to the opposite
study group for three additional months.
Fluorescein angiography showed no effect on CME, visual acuity, papillary
leakage, retinal vasculitis or choroidal leakage. A subgroup analysis
showed a decrease in both blood pressure and morning urinary albumin
excretion.
Source: van Kooij B, Fijnheer R, Joke de Boer J, et al. A
randomized, masked, cross-over trial of lisinopril for inflammatory
macular edema. Am J Ophthalmol 2006;141:646-651.
|